|
There are two unpaired electrons in the 2p subshell, so if carbon were to hybridize from this ground state, it would be able to form at most two bonds. The 1s electrons are considered to be core electrons and are not available for bonding. Let's look at the electron configuration of ground state (lowest energy state) carbon:įrom the ground state electron configuration, one can see that carbon has four valence electrons, two in the 2s subshell and two in the 2p subshell. By looking at the electron configuration, one is able to identify these valence electrons. The valence shell electrons are found in the incomplete, outermost shell. The hybridization of carbon involved in each of these bonds will be investigated in this handout.īonding in any element will take place with only the valence shell electrons. Carbon may form single, double and triple bonds. This is largely due to the types of bonds it can form and the number of different elements it can join in bonding. It may form virtually an infinite number of compounds. Hybrid orbitals normally participate in covalent sigma bond formation, whereas atomic orbitals participate in both sigma and pi bond formation.The element, carbon, is one of the most versatile elements on the periodic table in terms of the number of compounds it may form.But all the hybrid orbitals are equivalent and have the same electron number. Different atomic orbitals have different shapes and number of electrons.Different types and numbers of atomic orbitals are participating in making hybrid orbitals. 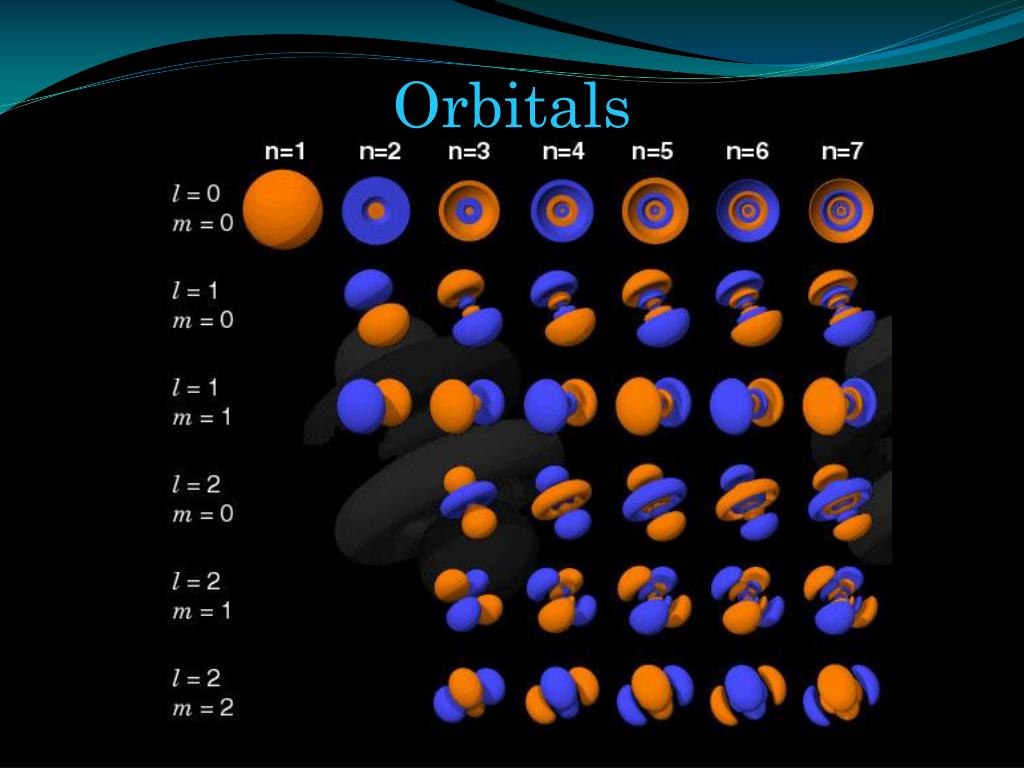
Hybrid orbitals are made from the atomic orbitals.What is the difference between Atomic Orbitals and Hybrid Orbitals? The number of hybrid orbitals produced is equal to the sum of orbitals being hybridized. Likewise in sp 2 hybridization three hybrid orbitals and in sp hybridization two hybrid orbitals are formed. Then the 2s electron and the three 2p electrons mix together and form four equivalent sp 3 hybrid orbitals. When excited, one electron in the 2s level move to the 2p level giving three 3 electrons. For example, in CH 4, C has 6 electrons with the electron configuration 1s 2 2s 2 2p 2 at the ground state. The most common hybrid orbitals are sp 3, sp 2 and sp. There are many types of hybrid orbitals formed by mixing s, p and d orbitals. The result of hybridization is the hybrid orbital. Hybridization is the mixing of two non equivalent atomic orbitals. So collectively, following are the total number of electrons that can be resided in an orbital. There are five d orbitals and 7 f orbitals with different shapes. They are arranged in space so that their axes are perpendicular to each other. The place where two lobes touch each other is known as a node. One lobe is said to be positive, and the other lobe is negative. P orbitals are dumbbell shaped with two lobes. The superscript number after the orbital name shows the number of electrons in that orbital. 1, 2, 3….n integer values are the quantum numbers. For example, when we write the electron configuration, we write as 1s 2, 2s 2, 2p 6, 3s 2. Atomic orbitals are characterized by quantum numbers, and each atomic orbital can accommodate two electrons with opposite spins. An atomic orbital can be defined as, a region of space where the probability of finding an electron is large in an atom. The plots of Ψ 2 in x, y, and z axes show these probabilities, and they take the shape of s, p, d and f orbitals. In contrary, if Ψ 2 is low, then the electron probability density there is low. Therefore, in the space, electron probability density is large. So, if Ψ 2 is a larger value, then the probability of finding the electron in that space is higher. 
According to Born, Ψ 2 expresses the probability of finding an electron in a particular location. Max Born points out a physical meaning to the square of the wave function (Ψ 2) after Schrodinger put forward his theory. Wave function (Ψ) corresponds to different states for the electron. 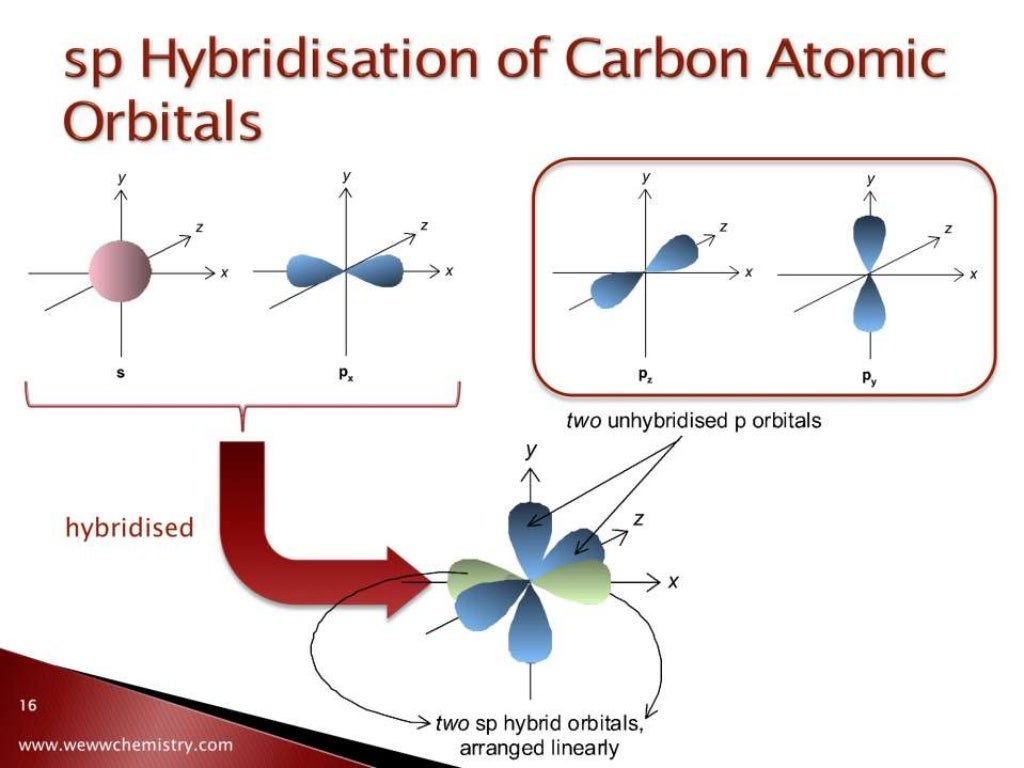
With this, Schrodinger developed equations to find the wave nature of an electron and came up with the wave equation and wave function. They found that an electron has both particle and wave properties. The quantum mechanics came into the picture with their findings. The bonding in molecules was understood in a new way with the new theories presented by Schrodinger, Heisenberg, and Paul Diarc.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
